We chose to look at building energy use from a thermodynamic point of view, in which we consider grades of energy to be as important as quantities of energy. Some of the grades at which energy exists in buildings are listed below (highest grade first).
1. Electricity for obligatory electricity users like electronic devices and motors. Gas and other fuels.
2. High temperature (> 100 C) heat for cooking.
3. Medium-temperature heat (50-70 C) in domestic hot water and wet central heating.
4. Low-temperature heat (20-30 C) in air.
5. Heat at ambient temperature.
The First Law of Thermodynamics tells us that energy cannot be created or destroyed. The practical consequence is that we can pass energy from one use to another in a building. That is, energy “used” on one rung of the “ladder” above can subsequently be “used” on another rung, but......the Second Law of Thermodynamics tells us that energy naturally degrades. The practical consequence is that we can only pass energy down the ladder above. (There is an exception which we can deal with later).
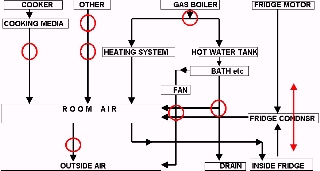
In the fairy-tale thermodynamically-efficient building we would put energy in at the highest grade and pass energy down the ladder, “using” it on every rung, before finally rejecting it to the environment in a useless form. In a practical building, we probably can’t achieve this performance. One reason is that there is a mismatch between the amounts of energy required on each rung of the ladder. For example, the amount of energy required for hot water will be much greater than the amount of energy used by obligatory energy users like televisions or washing machine motors. This mismatch means that we have to introduce energy at other stages of the ladder.
Guidelines for energy-efficient buildings
This line of thinking gives rise to some guidelines for energy-efficient building:
Strategy for designing the energy aspects of a building
Estimation of energy requirements
We estimated energy demands from the top down, starting with electricity, on the highest level. We aimed to use electricity only for jobs where there is no practical alternative.
We then went to the next level down, estimating how much total energy is required, and how much of this demand could be met by energy previously in the form of electricity. Thus we calculated the shortfall to be met by an external input.
We continued in this way, estimating, at each energy grade, how much energy is required, how much could be supplied by reject energy from higher grades, and how much needed to be supplied externally.
At the end of the process we end up with an estimate of external energy input required for space heating.
Possibilities for the reuse of energy
The thermodynamic fairy-tale building is probably not possible. For example, much obligatory electricity use, like radios and televisions, degrades the electrical energy straight to room temperature. The energy involved is dissipated in many small appliances and would be very hard to recover at a high temperature without great cost and restrictions on the design and mobility of appliances. However there are other, more profitable, opportunities.
1. Conventional buildings reject warm ventilation air to the environment, causing it to degrade to ambient temperature. By using a mechanical ventilation system with heat exchange, we can transfer this energy to incoming fresh air, minimising its degradation.
2. In a well-insulated building, the energy that exists in waste water (eg bathwater) is not negligible, and there may be ways to recover that energy for space heating before it degrades to ambient temperature.
Possibilities for the supply of energy
In practice, we are not surrounded by sources of energy at a range of grades. If our house is part of a CHP-driven district heating system, then some of our thermal energy needs can be met at an appropriate grade, but otherwise we are more or less compelled to use high grades of energy. This needn’t matter where the high-grade energy is abundant, for example, if we live in a very windy or a very sunny place, but if we are forced to use a scarce high-grade resource, we should do the best we can with it, thermodynamically.
One way of making appropriate use of high-grade energy is by heat pumping. Instead of degrading the high-grade energy directly to the level of use, we degrade it in a heat pump, which allows us to raise some very low-grade (and hitherto completely useless) energy up to the grade we want. In this way the “average” grade of energy supplied is roughly appropriate to the use to which we will put it.
Having found a way not to feel guilty about using high-grade energy for low-grade purposes, we need to find the most beneficial way of supplying it. In increasing order of preference, the sources are:
1. Nuclear or fossil-fuel electricity generated without use of reject heat.
2. Nuclear or fossil-fuel electricity generated with use of reject heat.
3. Renewably-generated electricity
Renewables
In an urban environment, we can most feasibly harvest solar power. The thermal use of solar power involves instant degradation of the energy from about 6000K to boiling point or less, but there’s not a lot that we can do about that. We need more energy at low grades than we need at high grades, and the energy itself (though not the equipment) is free, so the thermodynamic loss of opportunity isn’t too great. This argument applies to passive solar features like atria as well as dedicated solar thermal collectors.
PV systems allow us to convert the sun’s energy into a high-grade form to meet our high-grade uses. There is a potentially troublesome mismatch between time of supply and time of demand, and the enabling technologies of energy storage and demand management are important. Even here, we must think of energy grades, for some of the largest users of electricity (eg kettles, fridges) are the thermal uses, and it may turn out to be more practical to store the energy for these uses thermally rather than chemically.